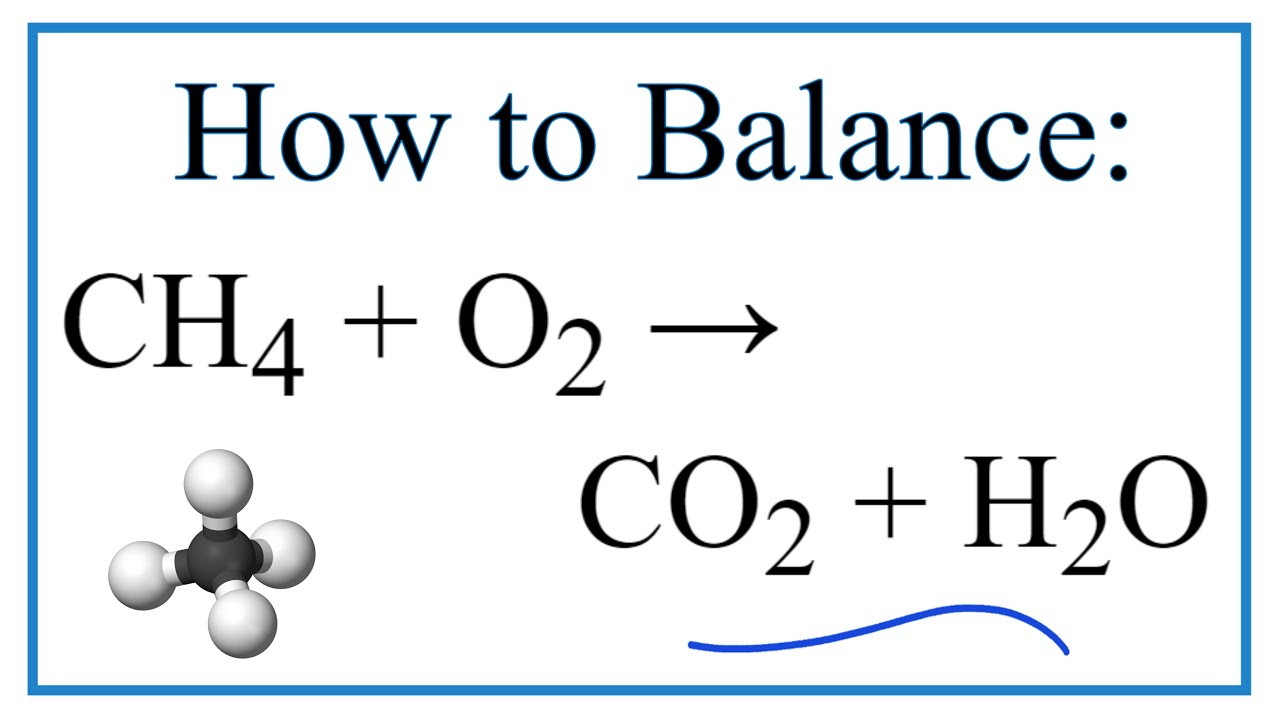 
The (aq) sign stands for aqueous in water and means the compound is dissolved in water. The (l) sign means the substance is a liquid. The (s) sign means that the compound is a solid. Often chemical equations are written showing the state that each substance is in. Since they undergo a chemical process, they are changed fundamentally. The equation shows that the reactants (AgNO 3 and NaCl) react through some process (->) to form the products (AgCl and NaNO 3). 
In this equation, AgNO 3 is mixed with NaCl. For example:ĪgNO 3(aq) + NaCl(aq) -> AgCl (s) + NaNO 3(aq) You will have a chance to review naming schemes, or nomenclature, in a later reading.Ī chemical equation is an expression of a chemical process. We call chlorine "chloride" in this case because of its connection to sodium. To represent a molecule of table salt, sodium chloride, we would use the notation "NaCl", where "Na" represents sodium and "Cl" represents chlorine. For example, the symbol "C"represents an atom of carbon, and "H" represents an atom of hydrogen. Successin chemistry depends upon developing a strong familiarity with these basic symbols. What is a chemical equation?In chemistry, we use symbols to represent the various chemicals. Given enough information, one can use stoichiometry to calculate masses, moles, and percents within a chemical equation. Chemical StoichiometryStoichiometry is the accounting, or math, behind chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
